Grignard Reaction
Grignard Reagents

The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or aldehyde, to form a tertiary or secondary alcohol, respectively. The reaction with formaldehyde leads to a primary alcohol.
Grignard Reagents are also used in the following important reactions: The addition of an excess of a Grignard reagent to an ester or lactone gives a tertiary alcohol in which two alkyl groups are the same, and the addition of a Grignard reagent to a nitrile produces an unsymmetrical ketone via a metalloimine intermediate. (Some more reactions are depicted below)
Mechanism of the Grignard Reaction
While the reaction is generally thought to proceed through a nucleophilic addition mechanism, sterically hindered substrates may react according to an SET (single electron transfer) mechanism: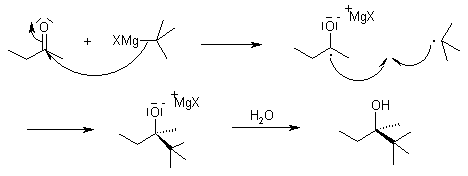
With sterically hindered ketones the following side products are received:
The Grignard reagent can act as base, with deprotonation yielding an enolate intermediate. After work up, the starting ketone is recovered.

A reduction can also take place, in which a hydride is delivered from the β-carbon of the Grignard reagent to the carbonyl carbon via a cyclic six-membered transition state.

Additional reactions of Grignard Reagents:
With carboxylic acid chlorides:

Esters are less reactive than the intermediate ketones, therefore the reaction is only suitable for synthesis of tertiary alcohols using an excess of Grignard Reagent:

With nitriles:

With CO2 (by adding dry ice to the reaction mixture):

With oxiranes:







0 Comments